How to Determine Limiting Reactant
A If the calculated MOLES NEEDED is greater than the MOLES HAVE for a given reactant then that reactant is the limiting reagent. To determine the limiting reactant following steps should be taken.

Chem 101 Dimensional Analysis Limiting Reagent Theoretical Yield Percent Yield Excess Reactant 2 Youtube
Remember to use the molar ratio between the limiting reactant and the product.
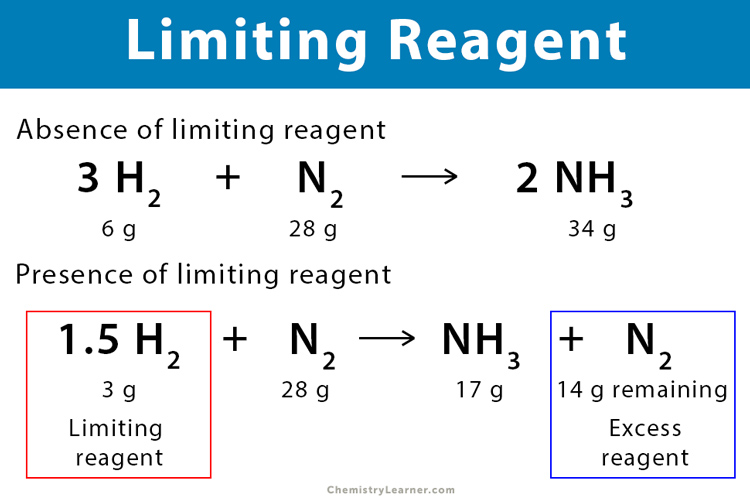
. An example is shown below-. Divide by the coefficients of the hydrogen. Now use the moles of the limiting reactant to calculate the mass of the product.
Calculate the number of moles H 2 given volume molar volume. How to Find the Limiting Reagent. Ad Over 27000 video lessons and other resources youre guaranteed to find what you need.
Determine the number of moles of each reactant. To determine which reactant is the limiting reactant first determine how much product would be formed by each reactant if all the reactant was consumed. Used to determine limitingexcess reagent.
Yes the percentage of yield can be calculated from the concept of limiting reactant. In this reaction ni 2 is the limiting reactant for the reaction. The limiting reactant or limiting reagent is the first reactant to get used up in a chemical reaction.
For the first method well determine the limiting reactant by comparing the. Find the limiting reagent by looking at the number of moles of each reactant. Get the number of moles of reacting substances from the given amounts of reactants.
If 25 ml of 0320 M barium chloride takes part in a reaction with. How To Determine Limiting Reactant Given Grams 2021 from wwwscottishindependencereferenduminfo Remember to use the molar ratio between the. This free limiting reactant calculator assists you to calculate limiting reactant that goes for finishing during the reaction and makes a limited amount of product.
Calculate the number of moles of. Divide the actual number of moles of each reactant by its stoichiometric coefficient in the balanced chemical equation. All of the methods give the same answer though so you can choose whichever approach you prefer.
Determine the balanced chemical equation. Causey shows you how to find the limiting reactant reagent and the maximum product from a chemical equation using stoichiometry. To determine how much product Fe 3 O 4 will be made multiply the limiting reactant times the mole ratio of product to the limiting reactant and then multiply by the molar.
We will work out a couple examples together and at the end of the. Lets go through the. Moles of HCl 025.
In this video we will learn how to determine the limiting reactant of a chemical reaction. Calculate the number of moles H 2 3022400.

How To Identify The Limiting Reactant In A Drawing Of A Mixture Chemistry Study Com

How To Find Limiting Reactant Quick Easy Examples Practice Problems Practice Questions Youtube

Limiting Reagent Reactant Definition Examples And Problems

Limiting And Excess Reactant Stoichiometry Problems Youtube

Limiting Reactant Practice Problems Youtube

0 Response to "How to Determine Limiting Reactant"
Post a Comment